How does behavior evolve?
The Johnson lab group is interested in the ecological factors that influence social behaviors and the physiological mechanisms that underlie those behaviors. Most of the lab’s work uses Caribbean lizards in the genus Anolis, or anoles, but we’re also exploring the diversity of lizards that occur at our local field sites in south-central Texas. We use field observations, laboratory experiments, molecular genetics, neuroendocrine techniques, and comparative methodology to explore behavioral evolution.
Click on the images below or scroll down to learn more about our research!
CURRENT PROJECTS
Evolution of Mechanisms of Behavior
One of the main avenues of research in the Johnson lab is the study of the muscular and hormonal traits that underlie behavior, and how those traits differ among species. In this work, we combine approaches from behavioral ecology and behavioral neuroscience to examine how variation in social behaviors is associated with variation in mechanistic traits.
We have focused much of this work on a group of 30 species of anoles, which exhibit two behaviors that are particularly interesting for this research: the display of a throat fan called a dewlap to communicate with other lizards, and the movement of the hemipenes during copulation.
We measure the frequency of dewlap extension and copulation behaviors in the field to determine how different species use these two behaviors (and the muscles that support them) at different rates, and then bring samples of muscle tissue into the lab. There, we study how muscles that are used differently differ in a wide range of physiological traits, such as cell size and number, androgen sensitivity, and enzyme function.
We also use this general approach to study the mechanisms of behavior in a wide diversity of lizards in Texas, including spiny lizards, whiptails, geckos, skinks, and earless lizards. In this work, we have examined how differences in lizard brain morphology are associated with modes of communication, how temperature influences brain and behavior, how lizards use their tails to communicate with predators and conspecifics, and how muscles and behavior change as a juvenile lizard develops into an adult.
This work has been funded by NSF IOS 1257021 to M.A. Johnson (2013-2018) and Texas Ecolab.
Representative publications:
Undergraduate coauthors indicated by *.
Johnson, M.A., R.E. Cohen, J. Wade. 2024. Neuroendocrinology of reptilian reproductive behavior. In: Hormones and Reproduction of Vertebrates, Volume 3, Reptiles, 2nd edition. D.O. Norris and K.H. Lopez, eds.
Johnson, M.A., B.K. Kircher, and D.J. Castro*. 2018. The evolution of androgen receptor expression and behavior in Anolis lizard forelimb muscles. Journal of Comparative Physiology A 204: 71-79.
Robinson, C.D.*, M.S. Patton*, B.M. Andre, and M.A. Johnson. 2015. Convergent evolution of brain morphology and communication modalities in lizards. Current Zoology 61: 281-291.
Communication with Color and Light
Another major theme of the research in the Johnson Lab involves how anole lizards use color to communicate with each other. Some of these studies have involved understanding how the dewlap - which can vary remarkably in color, pattern, and size, even within the same population of lizards - can send different information to other lizards. Other studies (in both the field and lab) have examined how anoles communicate with each other by changing their entire body color, often within seconds! In this work, we consider questions such as, do males and females use body color changes in the same ways? Is body temperature associated with body color? Does color communicate dominance or subordinance in a competitive interaction? Does parasite load influence body color in lizards?
In addition, we are studying how information from light determines how lizards interact with their environment. In collaboration with Dr. Simone Meddle from the Roslin Institute, University of Edinburgh, we have started to understand how lizards use opsin proteins - all over their body! - to detect light. We have also manipulated light exposure with artificial light at night, and by covering the parietal eye.
This work has been funded by the BBSRC to S. Mieddle and Texas Ecolab.
Representative publications:
Undergraduate coauthors indicated by *.
Horr, D.M.*, A.A. Payne*, K.D. McEntire, M.A. Johnson. 2023. Sexual dimorphism in dynamic body color in the green anole lizard. Behavioral Ecology and Sociobiology 77:34 10.1007/s00265-023-03308-8
Taylor, L.A.*, C.J. Thawley, O.R. Pertuit*, A.J. Dennis*, I.R. Carson*, C. Tang*, M.A. Johnson. 2022. Artificial light at night alters diurnal and nocturnal behavior and physiology in green anole lizards. Physiology & Behavior 257: 113992.
Bush, J.M.*, M.M. Quinn*, E.C. Balreira, M.A. Johnson. 2016. How do lizards determine dominance? Applying ranking algorithms to animal social behavior. Animal Behaviour 118: 65-74.
Cook, E.G.*, T.G. Murphy, M.A. Johnson. 2013. Colorful displays signal male quality in a tropical anole lizard. Naturwissenschaften 100: 993-996.
Reproductive Physiology
During the past several years, we have also been working to understand how variation in male and female reproductive tissues influence their behavior and physiology. Much of this recent work in our lab has focused on testis morphology, in collaboration with Dr. Ariel Kahrl of Hamilton College. We are examining, for example, how variation in testis architecture evolves in association with variation in sperm cell shape, and quantifying variation in these traits within species.
Representative publications:
Undergraduate coauthors indicated by *.
Kahrl, A.F., H.R. Hall*, I.R. Carson*, M.A. Johnson. 2025. The evolution of testis architecture and sperm size in Anolis lizards. Biological Journal of the Linnean Society 144: blaf016. https://doi.org/10.1093/biolinnean/blaf016
Martin, K.S., A.F. Karhl, B.M. Ivanov, M.A. Johnson. 2021. Use it and bruise it: Copulation rates are associated with muscle inflammation across anole lizard species. Journal of Zoology 314:187-193.
Johnson, M.A., M. V. Lopez*, T.K. Whittle, B.K. Kircher, A.K. Dill*, D. Varghese*, J. Wade. 2014. The evolution of copulation frequency and the mechanisms of reproduction in male Anolis lizards. Current Zoology 60:768-777.
Johnson, M.A. J. Wade. 2010. Behavioral display systems across nine Anolis lizard species: sexual dimorphisms in structure and function. Proceedings of the Royal Society of London: B 277:1711-1719.
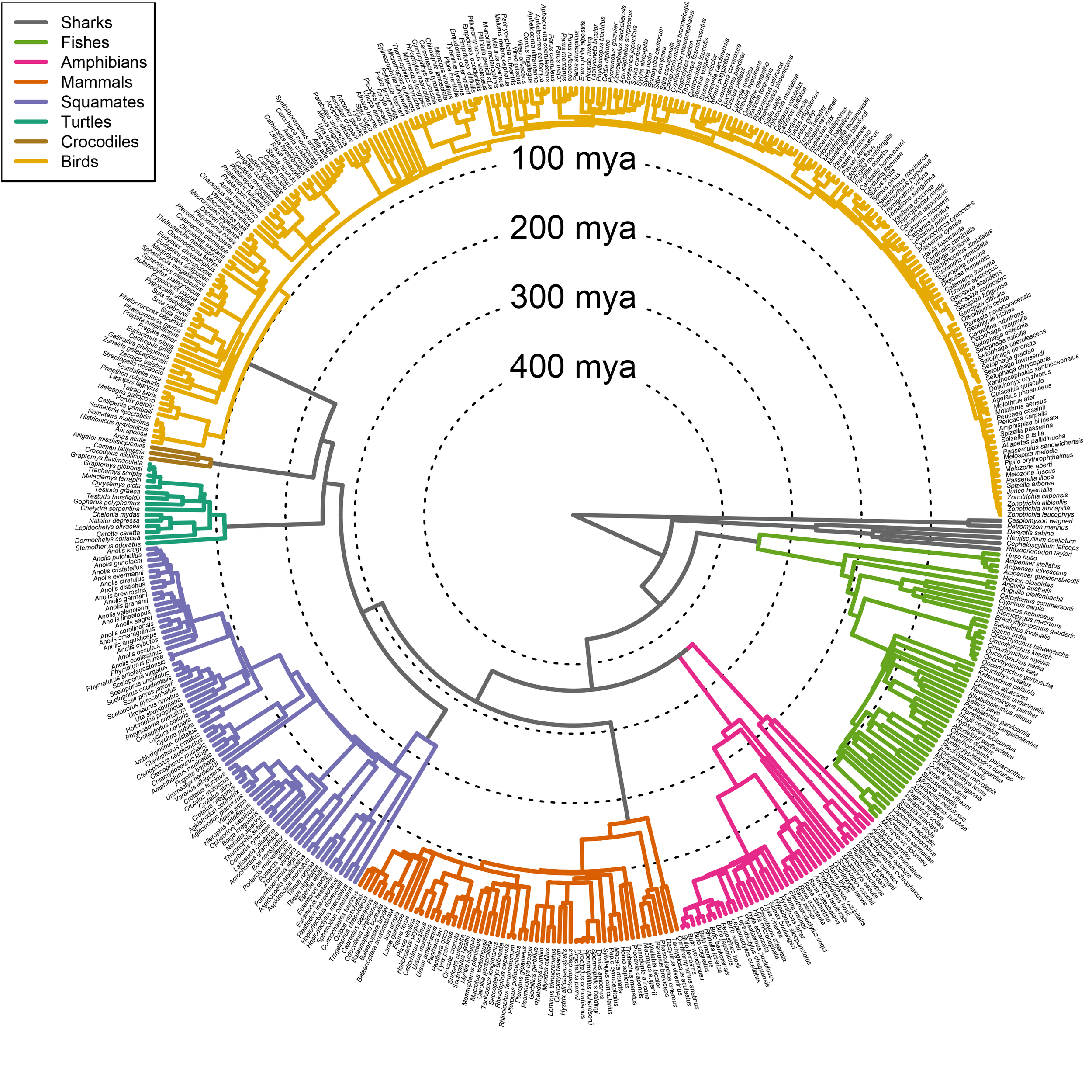
Comparative Databases
While many questions on the evolution of behavior can be answered using studies of individual species or species groups, larger scale questions require larger scale databases. We have been involved in compiling comparative databases, determining best practices for these efforts, and using the database, HormoneBase, to study patterns of hormone variation across vertebrates.
Dr. Maren Vitousek of Cornell University and Dr. Michele Johnson coordinated the HormoneBase Consortion to address questions of hormonal evolution. Our initial work required the compilation of a database of hormone measures, published in 2018 (freely available at hormonebase.org). The HormoneBase Consortium used this database to examine how circulating levels of androgens and glucocorticoids in free-living vertebrates are associated with life history traits, metabolism, speciation rates, and conservation status.
Representative publications:
Schwanz, L.E., A. Gunderson, M. Iglesias-Carrasco, M.A. Johnson, J.D. Kong, J. Riley, N.C. Wu. 2022. Best practices for building and curating databases for comparative analyses. Journal of Experimental Biology 8;225(Suppl_1):jeb243295. DOI: 10.1242/jeb.243295
McEntire, K., M. Gage, R. Gawne, M.G. Hadfield, C. Hulshof, M.A. Johnson, D.L. Levesque, J. Segura, N. Pinter-Wollman. 2021. Understanding drivers of variation and predicting variability across levels of biological organization. Integrative and Comparative Biology icab160. https://doi.org/10.1093/icb/icab160
Husak J.F, M.J. Fuxjager, M.A. Johnson, M.N. Vitousek, J.W. Donald, C.J. Francis, W. Goymann, M. Hau, B.K. Kircher, R. Knapp, L.B. Martin, E.T. Miller, L.A. Schoenle, T.D. Williams. 2021. Life history and environment predict variation in testosterone across vertebrates. Evolution 75:1003-1010.
Vitousek, M.N., M.A. Johnson, C.J. Downs, E.T. Miller, L.B. Martin, C.D. Francis, J.W. Donald, M.J. Fuxjager, W. Goymann, M. Hau, J.F. Husak, B.K. Kircher, R. Knapp, L.A. Schoenle, T.D. Williams. 2019. Macroevolutionary patterning in glucocorticoids suggests different selective pressures shape baseline and stress-induced levels. American Naturalist 193:866-880.
Johnson, M.A., C.D. Francis, E.T. Miller, C.J. Downs, and M.N. Vitousek. 2018. Detecting bias in large-scale comparative analysis: Methods for expanding the scope of hypothesis-testing with HormoneBase. Integrative and Comparative Biology 58:720-728.
Vitousek, M.N.,* M.A. Johnson*, J.W. Donald, C.D. Francis, M.J. Fuxjager, W. Goymann, M. Hau, J.F. Husak, B.K. Kircher, R. Knapp, L.B. Martin, E.T. Miller, L.A. Schoenle, J. Uehling*, and T.D. Williams. 2018. HormoneBase, a population-level database of steroid hormone levels across vertebrates. Scientific Data 5:180097. Asterisks indicate shared first authorship.